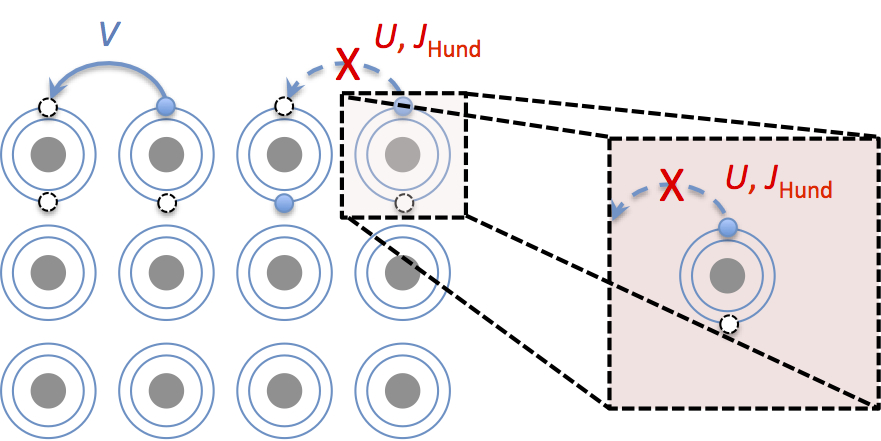
In atomic physics, Hund’s rules refers to a set of guidelines which are used to determine the term symbol that corresponds to the ground state of a multi-electron atom. The concept of a ‘Hund’s metal’ has arisen from the realization that the interactions that drive Hund’s rules for filling the orbitals of isolated atoms with electrons can also dramatically affect the hopping of electrons between atoms in solids, the motion of which is correlated and may support both high-temperature superconductivity and magnetism.
LCN researcher Dr. Cyrus Hirjibehedin has written a News & Views article providing an overview of this realization of the atomic-scale building block of a Hund’s metal. This follows a publication by Alexander Khajetoorians and colleagues in the same issue that reports that a single iron atom on a platinum surface forms the basic building block of a ‘Hund’s impurity’.
‘Magnetic atoms: The makings of a Hund’s metal’ can be accessed online in Nature Nanotechnology News and Views.